Diseases
- Human Health &Diseases
- Fundamentals of Health and Nutrition
- Understanding Diseases and Pathogens
- Major Communicable Diseases
- Major Non-Communicable and Genetic Diseases
- Emerging Diseases and Public Health Threats
- The Human Immune System
- Vaccination and Vaccine Technologies
- History of Diseases in India: From Ancient Times to the Modern Era
- Diseases Prelims Previous Year Questions
Vaccination and Vaccine Technologies
To fully understand public health initiatives, it is necessary to study the biological mechanisms behind vaccines and the different technologies used to manufacture them. This topic forms a crucial part of general science and technology.
Vaccination vs. Immunization
While these terms are often used interchangeably in general conversation, they have distinct medical meanings:
- Vaccination: This is the physical act of introducing a vaccine into the body (usually via an injection or oral drops).
- Immunization: This is the biological process by which a person’s immune system fortifies itself against a disease after receiving the vaccine. It is the end result of vaccination.
Vaccine
- The word “vaccine” has its origin in the Latin language. It comes from the Latin word “vaccinus,” which means “pertaining to cows” or “from cows.” This term was coined by Edward Jenner, an English physician, in the late 18th century.
- Edward Jenner observed that milkmaids who had been infected with cowpox, a disease similar to smallpox but milder, seemed to be immune to smallpox. In 1796, Jenner conducted an experiment in which he took material from a cowpox sore on a milkmaid’s hand and inoculated an 8-year-old boy named James Phipps. Later, he exposed the boy to smallpox and found that James did not develop the disease.
- Jenner named this procedure “vaccination” as a nod to the Latin term “vaccinus.” The use of the term “vaccine” has since evolved to refer more broadly to substances that stimulate the immune system to provide protection against various diseases, not just those related to cows.
- Vaccines typically contain various components that serve specific purposes. Here are some common ingredients found in vaccines:
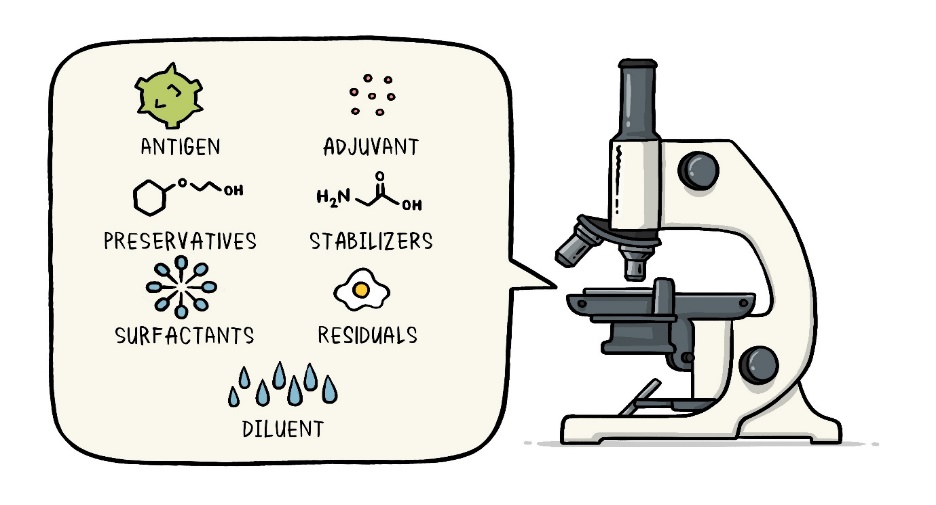
- Antigen: This is the active component of the vaccine that stimulates an immune response. It can be a small part of the disease-causing organism (like a protein or sugar) or the entire organism in a weakened or inactive form.
- Preservatives: These prevent contamination of the vaccine once the vial has been opened, especially if it will be used for vaccinating multiple people. Common preservatives include 2-phenoxyethanol.
- Stabilizers: Stabilizers prevent chemical reactions within the vaccine and prevent vaccine components from sticking to the vial. Examples include sugars (lactose, sucrose), amino acids (glycine), gelatin, and proteins.
- Surfactants: Surfactants keep all vaccine ingredients blended together and prevent settling and clumping. They are also used in foods like ice cream.
- Residuals: Residuals are tiny amounts of substances used during manufacturing that are not active ingredients in the vaccine. Examples include egg proteins, yeast, or antibiotics. Residual amounts are in such small quantities that they are measured as parts per million or parts per billion.
- Diluent: A diluent is a liquid used to dilute a vaccine to the correct concentration just before use. Sterile water is a commonly used diluent.
- Adjuvant: Some vaccines contain adjuvants to enhance the immune response. Adjuvants may include a tiny amount of aluminum salts.
The development of vaccines involves a meticulous process:
- Preclinical Phase: Screening and evaluations to determine the antigen, often conducted without testing on humans.
- Phase 1: Small-scale testing on young, healthy adult volunteers to assess safety, immune response, and dosage.
- Phase 2: Testing on several hundred volunteers to further assess safety, immune response, and various age groups.
- Phase 3: Testing on thousands of volunteers to determine effectiveness against the targeted disease and to study safety in a larger group. This phase is often conducted across multiple countries and sites.
After completing clinical trials, regulatory authorities review the data to decide whether to authorize the vaccine for use. Continuous monitoring of vaccine safety and effectiveness occurs even after a vaccine is introduced into the population.
