- Energy Resources
- Non-Conventional Sources of Energy
- Solar Energy
- Hydro energy
- Wind power
- Biogas
- Tidal Energy
- Geo Thermal Energy
- Nuclear or Atomic Energy
- Radioactivity
- Nuclear mechanism- Fusion & Fission
- Nuclear Reactor
- Fuelling a Nuclear Reactor
- Types of Nuclear Reactors
- Nuclear Energy in India
- India’s Three Stage Nuclear Program
- Innovative and Advanced Reactor Technologies in India
- Nuclear Fuel Cycle Facilities & Nuclear Waste Management
- Government Initiatives for Enhancing India’s Nuclear Capacity
- Advanced Energy Technologies & Storage
- Hydrogen Energy and Hydrogen Technology
- Fuel Cell
- Energy Storage Technologies
- Waste to Energy Plants
- Energy Security, Policies, and Government Initiatives
- Broader Energy Management & Grid Initiatives
- National Green Hydrogen Mission (NGHM)
- Green Energy Corridor
- Smart Meter National Programme (SMNP)
- Advanced Metering Infrastructure (AMI)
- Net Metering
- Pradhan Mantri Janjati Adivasi Nyaya Maha Abhiyaan (PM-JANMAN)
- Pradhan Mantri Sahaj Bijli Har Ghar Yojana (SAUBHAGYA)
- Production Linked Incentive (PLI) Scheme
- World Energy Investment Report 2025
- Electric Mobility Transition (FAME-1 &FAME-2)
- ENERGY RESOURCES Prelims Previous Year Questions
- Mains Previous Year Questions –ENERGY RESOURCES
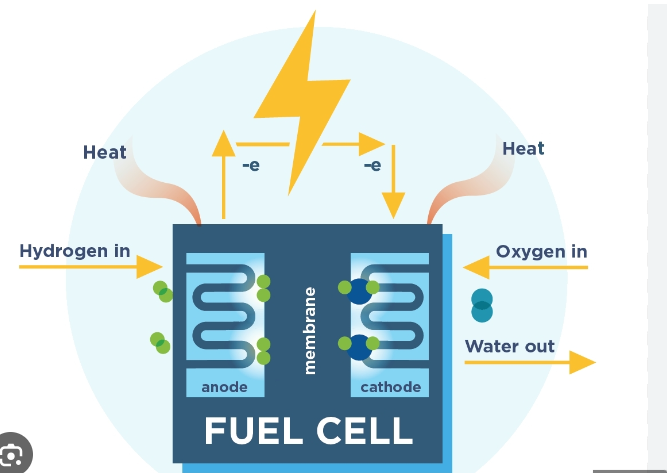
Fuel Cell
A fuel cell is an electrochemical device that converts the chemical energy of a fuel directly into electrical energy. Unlike conventional thermal power plants, a fuel cell does not first burn the fuel to produce heat and then convert that heat into electricity. Instead, it produces electricity through a controlled electrochemical reaction.
A fuel cell works somewhat like a battery, but there is one major difference:
- A battery stores energy within itself and gets exhausted after some time.
- A fuel cell continues to produce electricity as long as fuel and oxidant are supplied continuously.
Thus, a fuel cell is a continuous power-generating device.
Basic Components of a Fuel Cell
All fuel cells consist of three primary components sandwiched together:
1. Anode (Negative Electrode):
The terminal where the hydrogen fuel is introduced.
2. Cathode (Positive Electrode):
The terminal where oxygen (from the air) is introduced.
3. Electrolyte mebrane:
- It is the medium through which ions move from one electrode to another.
- It does not conduct electrons.
- The type of electrolyte determines the type of fuel cell.
2. Cathode (Positive Electrode):
- A catalyst speeds up the electrochemical reaction.
- Platinum is commonly used, though it is expensive.
- Scientists are developing cheaper alternatives.
5. External Circuit
- Electrons released at the anode travel through the external circuit to the cathode.
- This flow of electrons produces electricity.
The most common type used in vehicles today is the Proton Exchange Membrane (PEM) Fuel Cell, which operates at relatively low temperatures and is highly efficient.
The Electrochemical Reaction in Fuel Cell
The generation of electricity inside a hydrogen fuel cell involves two simultaneous chemical reactions.
At the Anode (Oxidation):
Hydrogen gas () enters the anode. A catalyst (usually platinum) causes the hydrogen molecules to split into positively charged protons () and negatively charged electrons ().
2H₂ → 4H⁺ + 4e⁻
2. The Flow of Energy:
The electrolyte membrane allows only the protons () to pass through the center to the cathode. The blocked electrons () are forced to travel through an external wire to reach the cathode. This flow of electrons through the external wire creates the electric current that powers the engine or device.
3. At the Cathode (Reduction):
Oxygen () from the air enters the cathode. Here, the oxygen combines with the protons that passed through the membrane and the electrons arriving from the external circuit to form pure water (H₂O) and heat.
O₂ + 4H⁺ + 4e⁻ → 2H₂O
Overall Reaction:
2H₂ + O₂ → 2H₂O + Energy + Heat